Authors: Ted Segarra, MD; Taylor Conrad, MD; Rithvik Balakrishnan, MD; Taylor M. Douglas, MD
Editors: Kami Hu, MD FAAEM and Kelly Maurelus, MD FAAEM
Originally published: Common Sense March/April 2019
Respiratory distress is a common presentation in both the pediatric and adult emergency department (ED). Community acquired pneumonia (CAP) is a common cause for this distress, and carries the potential for high morbidity and mortality if inadequately treated. In a fast-moving and potentially resource-limited ED, however, it can sometimes be difficult to decide which patients require further imaging to differentiate CAP from the myriad of other potential etiologies for respiratory distress such as bronchiolitis, asthma, chronic obstructive pulmonary disease (COPD), heart failure, and pulmonary embolism. Although both the British Thoracic Society (BTS) and Infectious Disease Society of America (IDSA) state that bacterial CAP is a clinical diagnosis based on persistent fever, retractions, and tachypnea, they agree that radiographic imaging should be obtained in any patient requiring hospital admission or with significant clinical uncertainty.[1,2] Unfortunately, even if the ED provider decides to pursue chest X-ray (CXR) imaging, he or she may still miss the diagnosis, as CXR has been shown in several studies to have a notable false negative rate (FNR) and high inter-observer variability in the diagnosis of CAP.[3,4,5,6] The limitations and inherent radiation exposure of CXR, in combination with the increasing availability of and familiarity with bedside lung ultrasound (LUS) imaging, have prompted many ED physicians to begin looking to LUS as a potential alternative in the evaluation of patients with suspected CAP.
- What is the level of sensitivity and specificity of LUS compared to traditional CXR and clinical findings in the diagnosis of CAP?
- Do other aspects of bedside LUS (i.e. lack of ionizing radiation, speed of assessment, easy repeatability, ability to monitor progression of disease, cost) make LUS a more feasible alternative in resource-limited environments?
Patel CJ, Bhatt HB, Parikh SN, et al. Bedside lung ultrasound in emergency protocol as a diagnostic tool in patients of acute respiratory distress presenting to emergency department. J Emerg Trauma Shock. 2018;11(2):125-9.
Studies have shown that ultrasound has the capability to diagnose pneumonia along with many other thoracic pathologies, and several researchers have worked to design and study a standardized approach to lung evaluation, especially in the critically-ill patient.[7] The bedside lung ultrasound in emergency (BLUE) protocol is an attempt at creating this standard approach that, in prior studies, has been shown to have a diagnostic accuracy of approximately 90%.[8,9] This process defines characteristics of different profiles that represent various disease states. Four anterior upper and lower left chest points are used, along with the posterolateral alveolar and/or pleural syndrome (PLAPS) point, which is essentially the posterolateral lung base. Several characteristic profiles are described and are listed in Table 1. The ultrasound findings that make up these characteristics include the following: A-lines, B-lines, absence or presence of lung sliding, lung point, C-lines or consolidated lung, PLAPS (presence of small pleural effusion and/or lung consolidation at the PLAPS point), and the presence of venous thromboembolism. The application and resulting pathologies associated are described in Figure 1.
| Table 1: The characteristics of each profile.[7] |
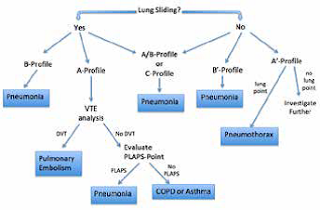 |
| Figure 1: BLUE Protocol starting with evaluation of the four anterior points. VTE is venous-thromboembolism. Adapted from Lichtenstein.[7] |
Patel, et al., attempted to replicate the prior BLUE protocol studies to determine the accuracy of the protocol when applied in the ED. This study was a small (n = 50), prospective observational study with patients over the age of 12 admitted to the intensive care unit (ICU) with acute respiratory distress. The patients were enrolled over a span of two months. The ED provider responsible for the ED care and disposition was the ultrasonographer; ICU teams were blinded to the ultrasound results and images. The prior mentioned BLUE protocol was utilized, and the patient’s ultimate diagnosis was compared to the initial presumptive diagnosis determined by the protocol. Non-standardized means of determining the final diagnosis were used and included patient history, treatment response, laboratory findings, X-rays and computed tomography (CT) scans. Specifically for pneumonia, when all profiles were considered, the authors report a sensitivity (Sn) of 94.1% and specificity (Sp) of 93.9%, corresponding to a positive likelihood ratio of approximately 15. The results were similar to prior findings by Lichtenstein and Dexheimer.[8,9]
This study is important in its attempt to replicate the results of previous studies and determine the protocol’s applicability in the ED. Numerous limitations exist, however, severely dampening any potential impact this study might have. The small size and fact that it is at a single center limits its generalizability. Additionally, the fact the ultrasonographer is also the treating ED provider introduces multiple biases and likely influences the future diagnosis, which in this study had no standardized method of being concluded. It is also important to note that only ICU patients were evaluated. Nevertheless, the bottom-line argument that LUS is an ever increasingly useful tool likely still holds. It is important to continue research into the utility of ultrasound and its application as a rule-in diagnostic tool for pneumonia and other lung pathologies.
Balk DS, Lee C, Schafer J, et al. Lung ultrasound compared to chest X-ray for diagnosis of pediatric pneumonia: a meta-analysis. Pediatr Pulmonol. 2018;53(8):1130-9.
Balk, et al., performed a systematic review and meta-analysis to compare the accuracy of LUS to CXR in the diagnosis of pediatric community acquired pneumonia (pCAP), searching PubMed, EMBASE, and Web of Science using the keywords “pneumonia,” “lung ultrasound,” and “pediatric population.” The authors required that these studies include pediatric patients, assess for bacterial pediatric CAP (pCAP), use both CXR and LUS, and use a gold standard of expert pediatrician clinical diagnosis for the ultimate diagnosis of pCAP. They initially identified 784 potential studies, which were then further screened and narrowed using Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) followed by a 14-item Quality Assessment of Diagnostic Accuracy Studies (QUADAS) tool to screen for relevant and unbiased studies. A final 12 studies including 1510 patients were ultimately selected for data extraction, and the authors used Freeman Tukey transformation of the data to calculate Sn, Sp, positive predictive value (PPV), and negative predictive value (NPV). Heterogeneity was assessed using Forest plots and calculations of inconsistency (I2) and Cochrane Q.
The 12 studies spanned 10 years (2008-2017) with a majority being prospective studies (9/12), half of which were from Italy with the remainder based in the USA, Turkey, India, Greece, Russia, and Taiwan. The study locations were equally split between the ED and inpatient floors. Four
studies relied solely on expert clinical diagnosis as the gold standard for pCAP, while the remaining eight studies used a combination of expert clinical diagnosis and CXR. In regard to the sonographic diagnosis of pCAP, all 12 studies used trained sonographers, eight of which used only experts. Of the remaining four studies, two did not specify sonographer experience level, one used novice radiologists, and one used both pediatric ED residents and expert sonographers. Ten studies used a 12-view lung evaluation, while the remaining two studies used an eight-view evaluation. LUS exams were considered positive for bacterial pCAP if they showed a) consolidation with or without air or fluid bronchograms (12 of 12 studies), b) focal B lines (4 of 12 studies), and c) pleural line abnormalities (1 of 12 studies).
The authors noted a significantly higher pooled Sn of LUS compared to CXR [95.5% (95% CI 93.6-97.1%) with no significant heterogeneity vs 86.8% (95% CI 83.3-90%) with significant heterogeneity]. Both LUS and CXR showed similarly high Sp (95.3% and 98.2%, respectively; with no significant heterogeneity between studies). There were no significant differences between LUS and CXR in PPV (99% and 99.6%, respectively) or NPV (63.1% and 43.6%, respectively).
Though overall a well-designed study demonstrating results consistent with prior literature, this meta-analysis contains a few notable limitations with regard to patient demographics and inter-study heterogeneity. All 12 study populations had a relatively high prevalence of disease and pretest
probability of pCAP, which likely explains the equally high PPVs and low NPVs across both study groups. This high prevalence of disease also limits extrapolation to populations without an equally high pretest probability of disease. In addition, since half of the studies were performed in Italy, the results may not be applicable to other patient demographics. Furthermore, there was notable heterogeneity between studies regarding both the clinical and sonographic diagnostic criteria of pCAP, which could skew the number of false positives and negatives. Despite these limitations, however, the authors found no significant heterogeneity in the final calculations of LUS Sn, LUS Sp, and CXR Sp, indicating that LUS may indeed be significantly more Sn in diagnosing pCAP, while sharing a similarly high Sp with CXR.
Amatya Y, Rupp J, Russell FM, et al. Diagnostic use of lung ultrasound compared to chest radiograph for suspected pneumonia in a resource-limited setting. Int J Emerg Med. 2018;11(1):8.
All studies prior to this one had been conducted in high- or mediumincome settings, and that factor spurred Amatya et al. to implement this method in Nepal. The importance of studying LUS in a resource-limited setting, in addition to the low generalizability of other research study results in this setting, is the truth that cheap, fast testing has a particularly important role when patients pay out-of-pocket and often must be transported by their family members for any tests ordered.
Criteria for inclusion in the study included age of 18 years or older with at least three of the following clinical characteristics: temperature >38°C, history of fever, cough, dyspnea, respiratory rate >20, and oxygen saturation <92%, in accordance with both the BTS and the European Society for Clinical Microbiology and Infectious Diseases (ESCMID) definitions of suspected CAP as well as criteria used in other studies.[2,10] After training physicians to perform LUS, they identified a convenience sample of patients with these characteristics whenever an ultrasound-trained physician was available. All enrolled patients received a Posterior-Anterior CXR as per usual care in this hospital and then also had a LUS exam and a CT scan of the chest (at no extra cost to the patient). Lateral views on X-ray were not included as they, in daily practice, incur an additional cost to the patient, further demonstrating the importance of cost in care in Nepal but also limiting the diagnostic value of the X-rays in the study.
A positive LUS exam was defined as the presence of unilateral B lines (A/B-profile) or subpleural lung consolidation (C-profile). Whether or not other BLUE protocol profiles were taught or evaluated is not mentioned, but these definitions are in accordance with the above and other BLUE protocol-based studies. Patients with positive radiologic exams were compared to patients with the same clinical characteristics who had negative radiologic exams. No comparison was made to patients who did not have the same clinical presentation, although this would be a valuable comparison due to the high prevalence of TB and chronic lung disease.
The incidence of pneumonia in the study, using CT as the diagnostic standard, was 71%, similar to rates seen in other studies. LUS demonstrated higher sensitivity for pneumonia than CXR (91 vs 73%, respectively, p=0.01) with no difference in specificity (61 vs 50%, respectively, p=0.62). Both CXR and LUS missed the same four cases of pneumonia, all in the middle of the lung parenchyma, deep to the pleura. The sensitivities of each modality are consistent with prior studies and meta-analyses in high-income and middle-income countries, however the specificities seen in this study are much lower, which the authors attribute to the higher prevalence of chronic lung disease and TB leading to more false positives. Importantly, LUS was faster (patients would not have to wait the two hours for transport, film acquisition, as well as preparation and interpretation of the image) and was performed at low/no cost to the patient, two factors that were not quantitatively measured but are inherent to LUS.
The major limitations of this study are its small size and inadequate power, convenience sample design, and lack of blinding of the ultrasonographers. It also left some questions unanswered regarding the design and results. One limitation in study design is that patients with these characteristics who presented when no trained physician was present were not identified or discussed, providing no evidence that this sample was not biased for an unidentified reason. Additionally, the lack of a lateral view potentially limited the performance of CXR, although inclusion of the lateral view would have increased cost to the patients, opposite the researchers’ goal. The authors also mention in the study protocol that if pneumonia was not clearly addressed in the radiologist interpretation then they were asked specifically to comment upon it. These studies could have been indeterminate or very obviously negative, however this distinction was not made or discussed so it is unknown if this would have affected results.
Notably, the study was not actually powered to evaluate specificity, so although there was no statistically-significant difference between the two groups, the outcome would perhaps change in a larger, appropriately powered study, and the authors also make a valid argument regarding false positives and subsequent lower specificity due to lung disease. They also admit their results are limited because they were in a single hospital in an urban area, but the same existing issues of out-of pocket costs, high pneumonia incidence, and high prevalence of chronic lung diseases and tuberculosis (TB) help make results seen in this setting applicable to other low-resource areas. Finally, one should consider the possibility that, in populations with a high prevalence of TB, it may be unwise to abandon CXR in favor of only LUS. This study, the first of its kind in a resourcelimited setting, provides a jumping-off point for future studies but does not represent definitive evidence that LUS for pneumonia will become the diagnostic method of choice in such areas.
Conclusion
While CAP and pCAP are considered clinical diagnoses, research and practice demonstrates that imaging is frequently used as an adjunct in cases with diagnostic uncertainty or unstable patients. LUS has most recently been proposed as a cheap, fast, bedside, radiation-free method to evaluate for CAP and pCAP. Various LUS criteria have been tested to determine what ultrasound characteristics should be used to diagnose CAP and pCAP, and the BLUE protocol has been demonstrated to be a well-defined set of criteria that could be used to standardize future research. The studies reviewed above present encouraging results, despite the heterogeneity and variability in clinical definition of CAP and pCAP, ability of ultrasonographers, and comparators (clinical decision vs CT vs CXR). The current research provides support for future studies into LUS as it compares to clinical decision-making, CXR, and CT as methods of identifying pCAP and CAP.
Now we return to the questions posed in the introduction:
1. What is the diagnostic accuracy of LUS compared to traditional CXR in the diagnosis of CAP?
The studies detailed in this article find LUS to have high sensitivity and specificity identifying CAP as compared to other modalities, such as CXR and clinical findings alone, but is dependent on sonographers’ level of experience and expertise. Specificity was lower in a resource-poor setting with high prevalence of other lung pathologies.
2. Do other aspects of bedside LUS (lack of ionizing radiation, speed of assessment, easy repeatability, ability to monitor progression of disease, cost) make LUS a more feasible alternative in resource-limited environments?
While these aspects of LUS make it an intuitively more appealing alternative on the surface, no current studies quantitatively assess them, and considerations have to be made regarding the need for well-trained ultrasonographers as well as the diagnostic value of the CXR in cases of TB, another lung disease prevalent in resourcelimited environment. More research is needed to fully evaluate the impact of LUS replacing CXR in this setting.
References:
1. Balk DS, Lee C, Schafer J, et al. Lung ultrasound compared to chest X-ray for diagnosis of pediatric pneumonia: a meta-analysis. Pediatr Pulmonol. 2018;53(8):1130-9.
2. Lim WS, Baudouin SV, George RC, et al. BTS guidelines for the management of community acquired pneumonia in adults: update 2009. Thorax. 2009;64:iii1-iii55.
3. Davies HD, Want EE, Manson D, et al. Reliability of the chest radiograph in the diagnosis of lower respiratory infections in young children. Pediatr Infect Dis J. 1996;15:600–4.
4. Swingler GH. Observer variation in chest radiography of acute lower respiratory infections in children: a systematic review. BMC Med Imaging. 2001;1.
5. Young M, Marrie TJ. Interobserver variability in the interpretation of chest roentgenograms of patients with possible pneumonia. Arch Intern Med. 1994;154:2729–32.
6. Maughan BC, Asselin N, Carey JL, et al. False-negative chest radiographs in the emergency department diagnosis of pneumonia. R I Med J. 2014;97:20–3.
7. Lichtenstein DA. BLUE-protocol and FALLS-protocol: two applications of lung ultrasound in the critically ill. Chest. 2015;147(6):1659-70.
8. Lichtenstein DA, Meziere GA. Relevance of lung ultrasound in the diagnosis of acute respiratory failure: the BLUE protocol. Chest. 2008;134(1):117-25.
9. Dexheimer Neto FL, Andrade JM, Raupp AC, et al. Diagnostic accuracy of the Bedside Lung Ultrasound in Emergency protocol for the diagnosis of acute respiratory failure in spontaneously breathing patients. J Bras Pneumol. 2015;41(1):58-64.
10. Woodhead M, Blasi F, Garau J, et al. Guidelines for the management of adult lower respiratory tract infections – Full version. Clin Microbiol Infect. 2011;17(6):E1-E59.

